COLONY MONTHLY ASSESSMENTS
|
The project is beginning this May and lasting through October 2015. Colonies will be split into two groups: a control group and a treatment group. Each beekeeper must have a minimum of one colony in each group. When choosing colonies for the study it is best if they are of similar strength and/or origin, for example the same queen breeder or of similar size. Control colonies are maintained through swarm preventative tactics and treatment colonies will be split in mid to late June. I will give a general date to split colonies and a colony split protocol to all participants. Throughout the field season (May–October), a total of six monthly colony assessments will be performed.
Monthly assessments are an evaluation of the strength and productivity of the colony and estimates of varroa mites. The strength of each colony is based upon five variables: adult bee and brood population, brood pattern quality, and the amount of pollen and carbohydrates (nectar and honey) in each colony. I have combined nectar and honey into one category when assessing the colony, carbohydrates (carbs). Adult bee and brood population and the amount of pollen and carbohydrates are measured by estimating its frame coverage to the nearest quarter, (0, ¼, ½, ¾ and 1). The quality of the colony’s brood pattern is based upon a 1-3 scale, 3 being the strongest and 1 being the weakest. Additional data recorded for each hive includes hive temperament, number of queen cups, queen status, varroa mite populations, symptoms of disease, and the presence of chalkbrood or American foulbrood. Colony temperament is estimated on a 1-3 scale, 1 being docile and 3 being aggressive. Common queen status’ include (1) present and laying, (2) no queen or (3) virgin queen. We are also assessing the status of the queen cups as: (1) queen cup(s), (2) charged with egg or larvae, or (3) capped queen cup. Colony assessments should be done once a month, about 30 day intervals, and it is important to wait at least one day in between varroa mite counts and colony assessments. Each colony assessment will take 30-60 minutes depending on the size of the colony and the time of season. Colonies that are assessed later in the season will probably be larger and take longer to assess. Each variable is described in detail below and I have included an example of the data entry sheet at end of the document. |

Procedures
During the project, uniformity in hive management between control and treatment colonies is important. Only treatment colonies will be split; however, to effectively test the efficacy of splitting colonies on varroa mite reductions it is important to minimize the variability of management practices between colonies. Below I have included guidelines to take into consideration when choosing colonies for the study and management practices.
During the project, uniformity in hive management between control and treatment colonies is important. Only treatment colonies will be split; however, to effectively test the efficacy of splitting colonies on varroa mite reductions it is important to minimize the variability of management practices between colonies. Below I have included guidelines to take into consideration when choosing colonies for the study and management practices.
- When choosing colonies for the study it is best if they are of similar strength and/or origin, for example the same queen breeder or of similar size.
- Colonies should be in a similar location so that environmental pressures and food and water availability is the same among all colonies.
- Management practices, beyond the splitting of colonies, should be practiced among all colonies. For example drone comb removal and feeding colonies.
- Varroa mite population estimates via sticky boards
- Symptoms of disease
- Hive productivity based upon estimating the amount of pollen and carbohydrates
- Adult bee and brood estimates
- Temperament and brood pattern quality
- Queen status
STEP 1: Specify brood box (shallow, medium or deep) and if it is a control or treatment colony
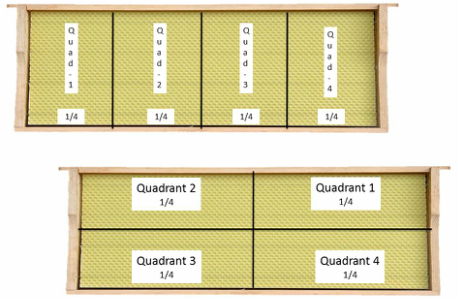
STEP 2: Split frame into quadrants
|
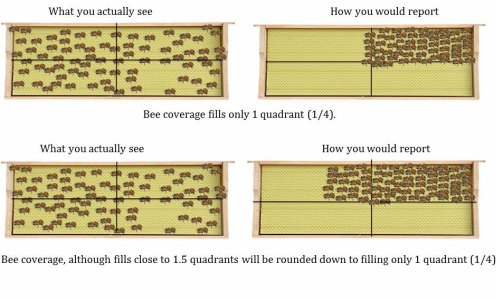
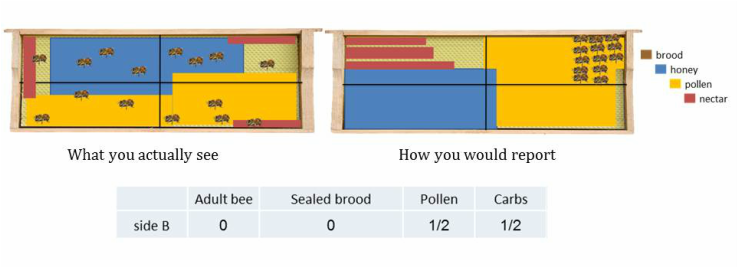
It is easiest to estimate frame coverage by splitting each side of the frame into quadrants. There are multiple methods to visually break the frame into quadrants; two are shown in the image to the left. Estimate the number of quadrants that each variable will fill (bees, brood, pollen and carbs) to nearest quarter (0, 1/4, 1/2, 3/4, 1). Be conservative on your estimates. Unless more than ¾ of a quadrant is filled it is rounded DOWN to the nearest quarter. Splitting the frame into quadrants will simplify the process of estimating frame coverage. |
Step 3: Estimate frame coverage of each variable
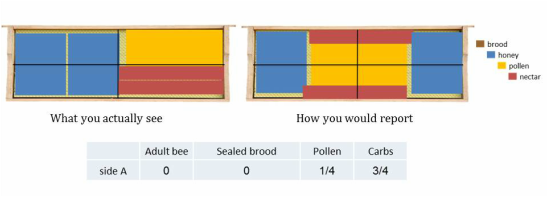
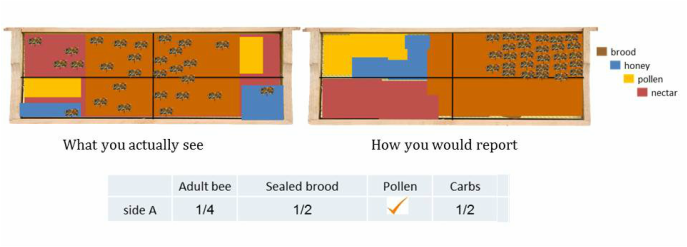
To accurately estimate percent frame coverage of each variable it is best to visually rearrange and group each variable on the frame. It is similar to piecing together a puzzle to determine how many quadrants each variable fills. In each example below there is a “what you actually see” and “how you would report” the variables on a frame.
STEP 3a: Estimate how many quadrants the bees will fill. It is important to condense the bees so there is minimal space between each bee.
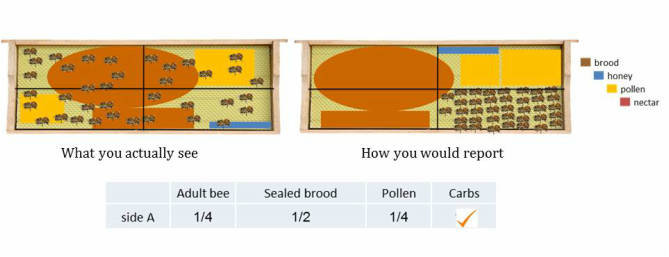
STEP 3b: Estimate the number of quadrants that brood, pollen and carbohydrates fill. This is different than estimating bee coverage because it is based upon cell coverage. Each variable should be grouped and condensed to where there are no empty cells. If there is pollen, carbohydrates or brood on the frame and it does not fill 1 quadrant than place a check mark in the box (not applicable to bees). Brood estimations does NOT include drone brood, only worker brood.
Not every frame is as straightforward and easy to estimate as the above examples. Many times the frame is a mosaic of variables arranged throughout the frame and this is when estimating becomes more difficult, as in the following examples.
STEP 4: Brood patterns
|
Brood pattern is NOT measured on a frame to frame basis or hive box. It is an overall estimate of the colony.
Brood guidelines 1. Brood pattern quality does not depend on the amount of the frame that is covered in brood, but on the total amount and consistency of the pattern of brood, larvae or eggs throughout the entire colony 2. The number of empty cells (not containing eggs, larvae nor brood) and the shape of the brood area define a pattern 3. The inner frames will have more brood typically than the outer frames that are reserved for pollen, nectar and honey 4. Brood pattern is based upon a scale of 1-3 5. A score of (3) will have minimal empty cells; a score of (1) the brood, eggs or larvae will be scattered and inconsistent STEP 5: Varroa mite measurements
|

The sticky board is the only mandatory monitoring method for measuring varroa mites. Sticky boards are an efficient method of estimating the total mite population within a colony and to monitor them over an extended period of time. There are two styles of sticky boards, full white and checkered sticky boards.
Varroa mite counts should be done at 30 day intervals. One day at minimum must be allocated between colony assessment and sticky board insertion because it will give the bees time to calm down.
The varroa mite is similar to a "flea on a bee".
Sticky boards can be purchased or homemade. It is a cut piece of cardboard paper to fit under the screen bottom board and covered with a sticky substance.
1. The sticky board is covered with a sticky substance (vegetable oil or petroleum jelly)
2. Insert the board sticky-side up below screen bottom boards or in the hive entrance with a screen cover on top to protect from bees for solid bottom boards
3. As mites are dislodged during grooming or removal from the capped cells they will fall through the screen cover and adhere to the board
4. The board is removed after 72 hours (3 days)
5. Mites are counted, although there could be debris on the board that could have to be sorted through when counting
1. The sticky board is covered with a sticky substance (vegetable oil or petroleum jelly)
2. Insert the board sticky-side up below screen bottom boards or in the hive entrance with a screen cover on top to protect from bees for solid bottom boards
3. As mites are dislodged during grooming or removal from the capped cells they will fall through the screen cover and adhere to the board
4. The board is removed after 72 hours (3 days)
5. Mites are counted, although there could be debris on the board that could have to be sorted through when counting
Step 6 Colony swarm prevention
Treatment colonies will not be split until mid-late June, I will provide a date as June approaches, and until then all colonies must be prevented from swarming. Swarming is related to space availability; here are several techniques that can keep the incidence of swarming low: 1). removing queen cups, charged and uncharged 2). providing ample room for queen egg laying and expansion of the brood nest 3). Providing ample room for pollen, nectar and honey storage.